संजीव दास
The focus of the laboratory is to understand the molecular mechanisms underlying the process of neoplastic transformation. The lab employs diverse approaches to examine the role of different regulatory networks and metabolic processes in cancer development and progression.
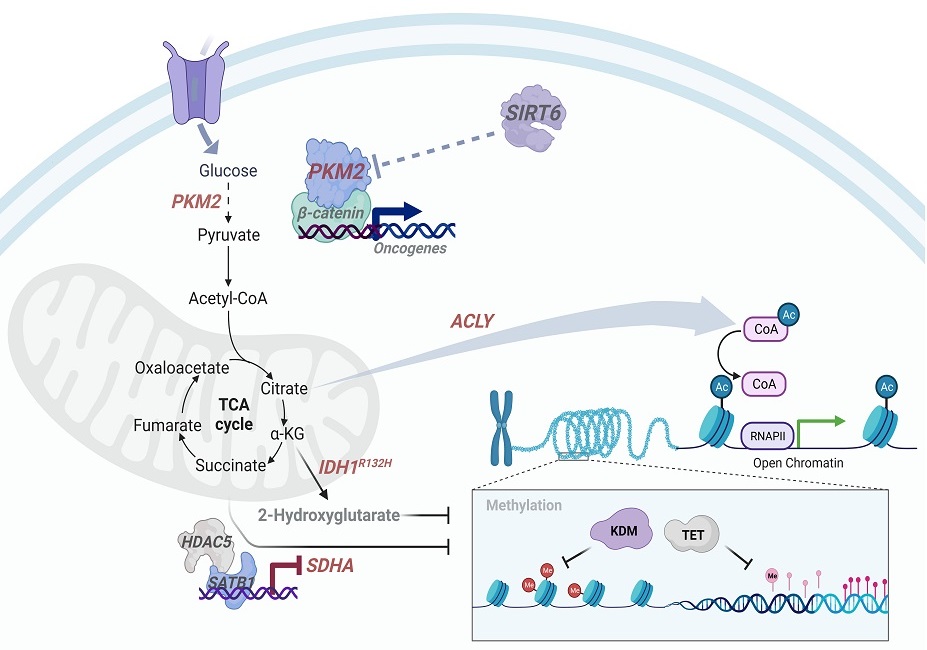
A complex disease such as cancer displays abnormalities of cellular metabolism, epigenome and transcriptome, and hence understanding the molecular perturbations underlying these processes will have vital therapeutic implications. The recent advances in understanding of tumor metabolism have provided an insight into the metabolic changes that augment proliferation, and, also the diverse mechanistic and context-dependent determinants that can trigger the metabolic reprogramming. The mechanisms underlying the communication between metabolic pathways and cell signalling networks are complex and have key roles in determining disease progression. Studies suggest that most metabolic pathways cross-talk with the machinery that controls gene expression. However, the molecular details of how the cytoplasmic signalling from metabolic state is conveyed to chromatin remodelling for congruent gene transcription remains poorly understood. The focus of our studies is to understand molecular perturbations that contribute to epigenomic-metabolic interplay in cancers. Using transcriptomics, RNA interference and proteomic screens we plan to delineate novel connections between the metabolic and the gene expression machinery. The current focus of the lab is on transcriptional regulators such as various HDACs and sirtuins which drive metabolic rewiring. Elucidating the role of metabolic enzymes such as PKM2, ACLY, SDHA and IDH1 which promote epigenetic plasticity underlying the malignant phenotype is also an area of notable interest in our research group. By gaining insights into these networks, we aim to create a foundation for tailoring precision therapeutic approaches.
Ph.D. Students: Anita Nandi, Mohd Irfan, Ricky Seldon Hemrom and Pawan Kumar Sharma
Technical Assistant: Rakesh Kumar
Awards:
• 2018 Fellow, National Academy of Sciences, India
• 2017 Shanti Swarup Bhatnagar Prize for Biological Sciences, Council for Scientific and Industrial Research, Govt. of India
• 2017 Prof. Umakant Sinha Memorial Award, Indian Science Congress Association, India
• 2016 Shakuntala Amir Chand Prize, Indian Council of Medical Research, India
• 2016 Prof. B. K. Bachhawat Memorial Young Scientist Lecture Award, National Academy of Sciences, India
• 2015 National Bioscience Award for Career Development, Department of Biotechnology, Govt. of India
• 2014 NASI-SCOPUS Young Scientist Award, National Academy of Sciences, India and Elsevier
- Tyagi W and Das S*. Temporal regulation of acetylation status determines PARP1 role in DNA damage response and metabolic homeostasis (2024). Science Advances, 10, eado7720.
- Madhurima Ghosh and Das S*. PRAMEF2-mediated dynamic regulation of YAP signalling promotes tumorigenesis (2021). Proc Natl Acad Sci USA, 118, e2105523118.
- Kumari R, Deshmukh RS and Das S*. Caspase-10 inhibits ATP-citrate lyase-mediated metabolic and epigenetic reprogramming to suppress tumorigenesis (2019). Nature Communications, 10, 4255.
- Deshmukh RS, Sharma S and Das S*. Cyclin F-dependent degradation of RBPJ inhibits IDH1R132H-mediated tumorigenesis (2018). Cancer Research, 78, 6386-6398.
- Kohli S, Bhardwaj A, Kumari R and Das S*. SIRT6 is a target of regulation by UBE3A that contributes to liver tumorigenesis in an ANXA2-dependent manner (2018). Cancer Research, 78, 645-658.
- Bhardwaj A and Das S*. SIRT6 deacetylates PKM2 to suppress its nuclear localization and oncogenic functions (2016). Proc Natl Acad Sci USA,113, E538-E547.
- Satija YK and Das S*. Tyr99 phosphorylation determines the regulatory milieu of tumor suppressor p73 (2016).Oncogene,35, 513-527.
- Sen N, Kumari R, Singh MI and Das S*. HDAC5, a key component in temporal regulation of p53-mediated transactivation in response to genotoxic stress (2013). Molecular Cell,52, 406-420.(Featured Article)
- Sen N, Satija YK and Das S*. PGC-1a, a key modulator of p53, promotes cell survival upon metabolic stress (2011). Molecular Cell,44, 621-634.(Featured Article)
* as corresponding author



























